Ionization, Entropy, and Dark Matter
Below is a post from Gene Ellis. I don’t necessarily agree or disagree with his views, but CNPS welcomes new ideas on a wide variety of science topics, so please comment and let us know your thoughts.
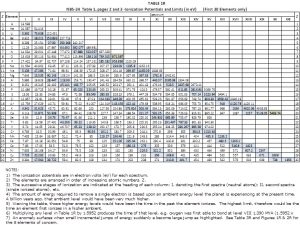
Energy level data was compiled in 1970 by Charlotte E. Moore; NBS-34, Ionization Potentials and Ionization Limits Derived from the Analysis of Optical Spectra. The defined ionization potential of an element is a measure of its ability to enter chemical reactions requiring ion formation or donation of electrons and is related to the nature of the chemical bonding in compounds formed by elements. Ionization, however, encompasses more than this definition. The ionization potentials and limits shown in Table 1 are energy levels in electron volts that correspond to the electron activity of the elemental atoms. One electron volt (1eV) is the amount of energy an electron gains moving through a potential of one volt in a vacuum. For each element, the largest number in each row is the energy limit in electron volts. When reading the chart from right to left (from past to present) it marks the first time the element is able to enter a reaction or bond. Prior to the time of this first ionization, the element is neutral because no electrons are detected or interacting as an electromagnetic force. Since the mass of an element is unchanging before the time of its highest ionization (potential) and the presence of such masses are not analytically revealed, those masses would be classified as dark matter.
The time an element first ionizes raises another question…whether the electrons appear singularly at each ionization level or all at once at the highest level? Resolution of this “unknown” could alter our present concept of the quantum-mechanical model of the atom.
An anomaly is noted when the small incremental jumps between energy levels become large jumps as highlighted in Table IR. These large jumps create a pattern and indicate something different occurring within the atoms. These occurrences, as presented in the IGE 8-Element Supplement, are attributed to entropy for when the elemental atoms are heating.
Knowing the time as related to the spectrum, changes what we think we know about our planet and the universe. Before calcium ionized for the first time ~8800 MYA, all elemental matter was dark matter. Depending upon how electrons emerge, water did not exist until oxygen ionized either ~1400 MYA or ~1200 MYA. Hydrocarbons probably did not arrive until ~800 MYA and all hydrogen existed as dark matter until ~ 22 MYA when it ionized for the first time and could join to become a molecule (H2).
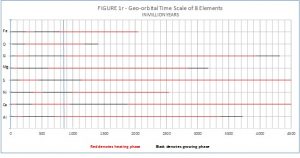
When the growing and heating phases of the eight elements that comprise 98.8% of Earth’s matter are plotted as in Figures 1R and 2R, it is obvious the planet was primarily heating for the first 3000-3500 million years of its existence. During this time there was very little change in the size of the planet. Molten crustal rock solidified after water arrived as the cooling agent ~1200 or ~1400 MYA. Consequently, radiometric dating only extends back to such time when rock solidified. The chart also indicates exponential growth essentially beginning ~700-800 MYA.
All of the above result from when the energy decay rate (1eV/1.6 MY) is applied to the Ionization Potentials and Ionization Limits of NBS-34 and illustrates:
Ionization is a mechanism conditionally allowing elements to join at certain energy levels.
Ionization is the mechanism that emerges the electrons to make dark matter spectra visible.
Ionization also is a switch-like mechanism that directs energy to convert to mass or to entropy that grows and heats elemental atoms.
Earth’s internal heat from radioactive decay is an addition to the heat from elemental decay.






Dear Gene
I apologize that I am not better informed on what you discuss. I assume that you consider the Earth’s expansion has little to do with accretion. I am not sure what to say concerning the theory
I will say this:
In your papers you use the term entropy a fair bit. Do you mind me asking what is your definition or understanding of entropy in your theory?
I also want to state: You discuss the second law in one of your papers and say that it explains why heat always irreversibly flows from hot to cold. Me I am not particularly enthusiastic about such considerations. Our reality is that the exchange of thermal energy (AKA heat) goes from cold to hot as it does from hot to cold. It is just that the amount of thermal energy transferred from a hot body to a cold body is always greater than that from a cold body to a hot body. In other words the net flow of thermal energy that we can measure is always from hot to cold. This has nothing to do with entropy or the second law. It is just plain simple logic that the net flow is from the highest concentration/density of thermal energy to the lowest. And yes the flow of heat between two bodies at different temperatures is irreversible but that too should be based upon simpleton logic.
As for the rest of it I will try to arrive at a better understanding. I should actually have more interest in this than I currently do as my background is really geophysics based.
All the best Kent
Hello Kent,
Your assumption is correct…Earth’s expansion has very little to do with accretion. The quantities required for doubling the size of the planet are too great for accretion to be possible.
As you say in your A NEW BEGINNING paper…”entropy has different interpretations, all dependent upon it application.” Here, entropy is viewed and not thought of as a form of energy but as a separate and independent entity of heat and temperature. Work can be extracted from heat and, as you indicate, the net effect is a loss of heat. From this, the question becomes…where does the lost heat go? Logically, it ends up in space with all the other heat generated from the billions of stars for billions of years. An equally important question is…where does the heat originate? Heat sufficient to raise the temperature of the early Earth above the melting point of the iron that gravitated to the core. Also, where does the pressure originate within the Earth (when combined with the heat) that causes volcanism and separation of the tectonic plates.
The answers seem to flow from analyzing the ionization data of the eight elements that comprise 98.8% of Earth’s matter. The key is establishing an energy decay rate of 1eV/1.6 MY that reveals the time when the planet is heating and when it is growing (expanding). It also reveals when water arrived and why it also is doubling, and filling the oceanic basins as the planet doubles in size. Most surprising is the time when elemental dark matter becomes spectra visible (detectable). When Earth coalesced ~4.5 BYA, less than 4.5% of its matter was spectra visible. The remaining 95.5% existed as dark matter to provided sufficient mass at a reasonable distance from the Sun (per Newton). In modern physics, dark matter was assumed to exist to satisfy Newton’s gravity, but none could be found. Presumably, all Earth’s elements have ionized; therefore no elemental dark matter is currently present on this planet. I must admit…this evidence changed my thinking about dark matter.
Incidentally, I have a few corrections to the tables of the Growing Earth / Expanding Universe (GE/EU) paper presented at the CNPS-2016 Conference last July. A corrected version should be on my website next week.
I think I answered your question with more questions. Thank you for your interest.
Gene